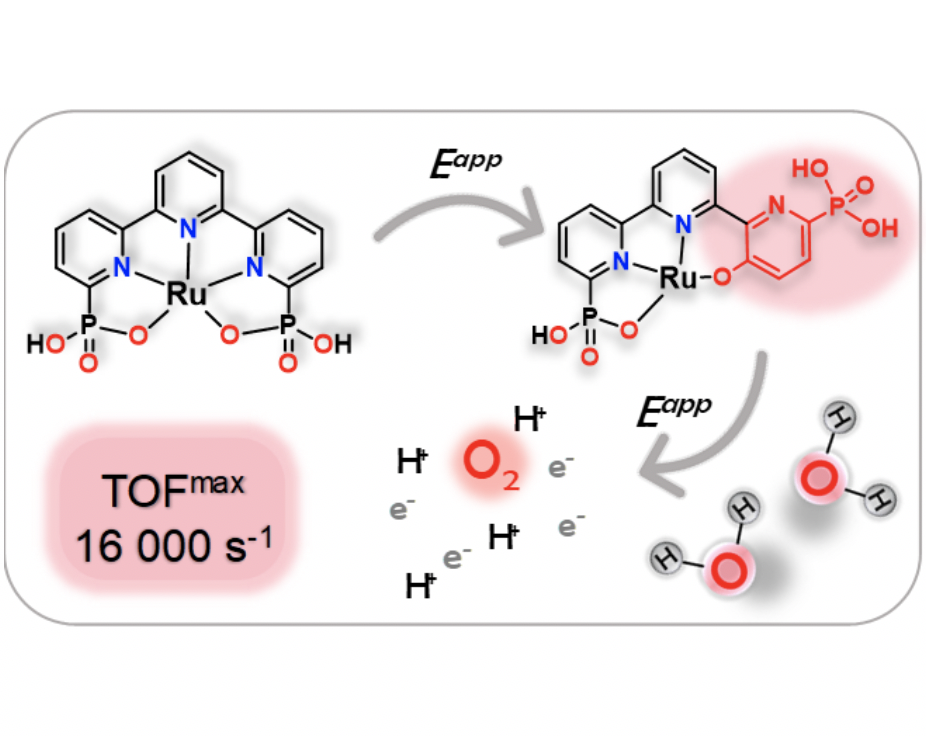
A new Ru complex containing the deprotoanted 2,2′:6′,2”-terpyridine,6,6”-diphosphonic acid (H4tPa) and pyridine (py) of general formula, [RuII(H3tPa--N3O)(py)2]+, 2+, has been prepared and thoroughly characterized by means of spectroscopic, electrochemical techniques, X-ray diffraction analysis and with DFT calculations. Complex 2+, presents a dynamic behavior in solution that involves the synchronous coordination and the decoordination of the dangling phosphonic groups of the tPa4- ligand. However, at oxidation state IV complex 2+, becomes seven coordinated with the two phosphonic groups now bonded to the metal center. Further at this oxidation state at neutral and basic pH, the Ru complex undergoes coordination of an exogenous OH- group from the solvent that leads to an intramolecular aromatic O-atom insertion into the CH bond of one of the pyridyl groups forming the corresponding pheoxoxo-phosohonate Ru complex [RuIII(tPaO--N2OPOC)(py)2]2, 42, where tPaO5- is 3-hydroxo-[2,2′:6′,2”-terpyridine]-6,6”-diyl)bis(phosphonate) ligand. This new in situ generated Ru complex, 42-, has been isolated and spectroscopically and electrochemically characterized. In addition, a crystal structure has been also obtained using single crystal X-ray diffraction techniques. Complex 42-, turns out to be an exceptional water oxidation catalyst achieving record high TOFmax in the of 16,000 s-1. A mechanistic analysis complemented with DFT calculations has also been carried out showing the critical role of intramolecular second coordination sphere effects exerted by the phosphonate groups in lowering the activation energy at the rate determining step.