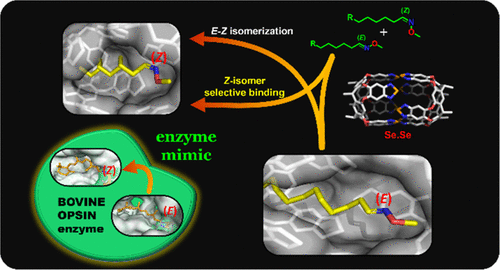
A series of straight-chain (C7–C13) alkyl-O-methyl aldoximes (R–C(H)═NOMe) were synthesized with various functional groups at the remote ends (alkenes, halogen, −COOH, and NH2). Their isomers about the C═N bond showed ∼60–40% E–Z-ratio in organic solutions. Surprisingly, their confinement in a water-soluble capsule with benzoselenodiazole walls shows high selectivity for the cis-/Z-isomer. Their relative affinities for the chalcogen-bonded capsule at room temperature depend mainly on the guest chain length and functional groups. A chain length of 14 heavy atoms showed especially high E- to Z-isomer selectivity (>99%) and was used in separation. The E–Z isomerization occurred only in the capsular cavity at room temperature and was accelerated 10-fold by sonication. The Z-isomer selective binding, separation, and E–Z isomerization are supported by NMR, DOSY, and computational studies.