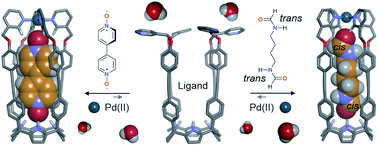
Coordination cages containing endohedrally functionalized aromatic cavities are scarce in the literature. Herein, we report the self-assembly of a tetra-cationic super aryl-extended calix[4]pyrrole tetra-pyridyl ligand into a water-soluble Pd(II)-cage featuring two endohedral polar binding sites. They are defined by the four pyrrole NHs of the calix[4]pyrrole unit and the four inwardly directed α-protons of the coordinated pyridyl groups. The efficient assembly of the Pd(II)-cage requires the inclusion of mono- and ditopic pyridyl N-oxide and aliphatic formamide guests. The monotopic guests only partially fill the cage’s cavity and require the co-inclusion of a water molecule that is likely hydrogen-bonded to the endohedral α-pyridyl protons. The ditopic guests are able to completely fill the cage’s cavity and complement both binding sites. We observed high conformational selectivity in the inclusion of the isomers of α,ω-bis-formamides. We briefly investigate the uptake and release mechanism/kinetics of selected polar guests by the Pd(II)-cage using pair-wise competition experiments.