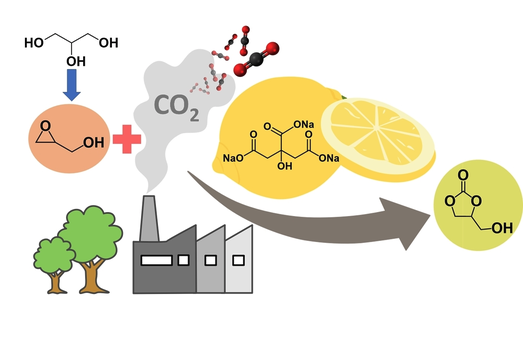
Glycerol carbonate (GC) has emerged as an attractive synthetic target due to various promising technological applications. Among several viable strategies to produce GC from CO 2 and glycerol and its derivatives, the cycloaddition of CO 2 to glycidol represents an atom-economic an efficient strategy that can proceed via a halide-free manifold through a proton-shuttling mechanism. Here we show that the synthesis of GC can be promoted by bio-based and readily available organic salts leading to quantitative GC formation under atmospheric CO 2 pressure and moderate temperatures. Comparative and mechanistic experiments using sodium citrate as the most efficient catalyst highlight the role of both hydrogen bond donor and weakly basic sites in the organic salt towards GC formation. The citrate salt was also used as a catalyst for the conversion of other epoxy alcohols. Importantly, the discovery that homogeneous organic salts catalyze the target reaction inspired us to use metal alginates as heterogeneous and recoverable bio-based catalysts for the same process.