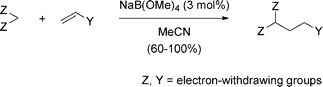Sodium tetramethoxyborate, easily prepared by reaction of inexpensive sodium borohydride with methanol, possesses a suitable combination of a Lewis base and a Lewis acid to catalyze Michael reactions at room temperature under practically neutral conditions. This reaction provides good to excellent yields of Michael addition products from a broad scope of Michael donor and Michael acceptor reagents.