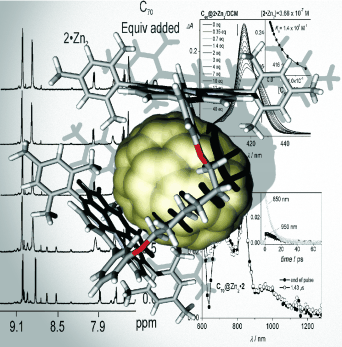
The formation of thermodynamically stable inclusion complexes between two cyclic zinc bisporphyrins, differing in the saturation degree of the hydrocarbon linkers that connect their porphyrin units, and the fullerenes C60 and C70 is described. Binding and photophysical studies were performed in two solvents of very different polarity: toluene and dichloromethane. UV/Vis and fluorescence titration experiments showed π-π interactions between the cyclic zinc bisporphyrins and the fullerenes. Solid-state structures were determined by X-ray diffraction analysis and gave valuable insight into the different complexation behaviors of the two macrocyclic systems towards the fullerenes. NMR titrations were also helpful in understanding the geometry of the complexes in solution. Upon fullerene complexation, the two macrocyclic bisporphyrins adopt very distinct conformations. Charge-transfer absorption bands point to ground-state interactions, and quenching of the porphyrin component luminescence indicates fast reactivity in the excited states. Energy transfer plus HOMO-HOMO and LUMO-LUMO electron-transfer processes occur within the complexes. Charge-separated states characterized by a reduced fullerene and an oxidized porphyrin radical, with lifetimes in the order of several hundred picoseconds, are detected.