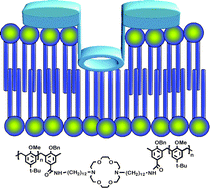
An earlier study showed that a calix[4]arene could function as a central relay unit to form an ion conductance pathway through a phospholipid bilayer membrane. The present study expands the range of compounds from calix[4]arene to calix[6]arene and incorporates them either as central units or as headgroups, substituting one or more diaza-18-crown-6 residues in functioning hydraphiles. Ion release was assayed by detecting either Na+ or Cl– release from phospholipid vesicles. The ion transport activity for calix[4]arenes in either position is modest, but is almost non-existent when calix[6] residues were incorporated either as head groups or central relay units. The poor activity of the calix[6]arenes may result from an inability to penetrate to the midplane of the bilayer or pass entirely through it to form a conductance pathway. The transmembrane flip-flop may result from high polarity or steric bulk, or both. A hydraphile incorporating a single -NHCOC6H4OCH2CONH- as a central relay proved to be an excellent Na+ conductor, but less selective for Cl–. The fact that this new hydraphile molecule shows selectivity for Na+ over Cl– transport and possesses two secondary amide residues in the central relay suggests a means to control ion selectivity in synthetic ion transporters.