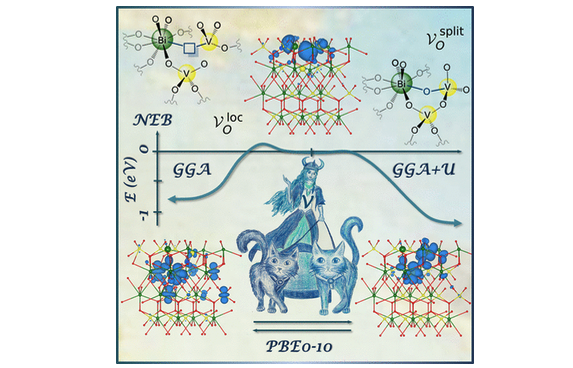
Bismuth vanadate (BiVO4) has emerged as one of the most promising photoanodematerials for solar fuel production. Oxygen vacancies play a pivotal role in the photoelectrochemical efficiency, yet their electronic nature and contribution to the type conductivity are still under debate. Using first principles calculations, we show that oxygen vacancies in BiVO4 have two distinguishable geometric configurations characterized by either undercoordinated, reduced V(IV)O3 and Bi(II)O7 subunits or a V(IV)–O–V(IV)/Vbridge (split vacancy), quenching the oxygen vacant site. While both configurations have similar energies in the bulk, the (001) subsurface acts like an energetic sink that stabilizes the split oxygen vacancy by∼1 eV. The barrierless creation of a bridging V2O7unit allows for partial electron delocalization throughout the near surface region, consistent with recent experimental observations indicating that BiVO4(001) is an electron rich surface.