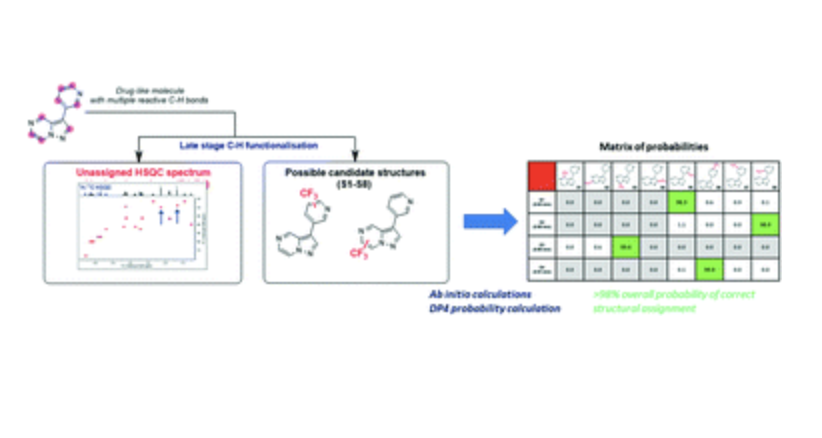
An automated analysis pipeline is outlined that helps unlock the true potential of late stage functionalisation (LSF). LSF is an extremely powerful strategy, of increasing importance, for the rapid expansion and exploration of structure-activity relationships in life science applications. However, broad use installing various groups on multiple parent compounds shifts the bottle-neck from synthesis to the elucidation of product structure. To address this key limitation, we have developed a high-throughput, automated and reliable methodology for product structure determination. This approach is based on chemical shift calculations and probabilistic DP4 analysis. It uses easily generated, unassigned HSQC data and can easily accommodate other data from NMR or other techniques within its scheme and provides a significant advance in unlocking the potential of LSF for rapid SAR exploration.. This method has been applied to the products of LSF reactions, regioisomeric compounds from our compound collection and for the mapping of regioselectivity in LSF reactions, from which its accuracy and utility is assessed.