Abstract
Understanding reaction mechanisms is the key to developing new chemical reactions. Electrospray ionization mass spectrometry has a unique dynamic range that allows studying reaction mixture compositions, including low abundant reactive intermediates. In the lecture, I will show several approaches to studying reaction intermediates in small-molecule activation reactions,[1-3] including their spectroscopic characterization by vibrational and electronic spectroscopy.[4]
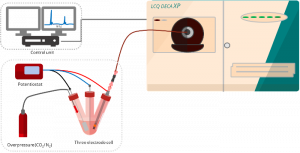
The strategy for small molecule activation (N2, O2, CO2) often includes electrochemical reduction reactions. We have developed an approach to bridge voltammetry experiments with screening species formed at the electrode by electrospray ionization mass spectrometry. Our setup lets us couple the redox waves in a cyclic voltammogram with detected ions. An example of monitoring intermediates in oxygen reduction will be discussed.
References
[1] J. Mehara, J. Roithova, Identifying reactive intermediates by mass spectrometry, Chem. Sci. 11 (2020) 11960.
[2] G. L. Tripodi et al., Tracking Reaction Pathways by a Modular Flow Reactor Coupled to Electrospray Ionization Mass Spectrometry, Chem. Methods 1 (2021) 430.
[3] A. Koovakattil Surendran et al., Host-guest tuning of the CO2 reduction activity of an iron porphyrin cage, Nat. Sci. 3 (2023) e20220019.
[4] J. Roithová et al., Helium Tagging Infrared Photodissociation Spectroscopy of Reactive Ions, Acc. Chem. Res. 49 (2016) 223.